Abstract
Chemical descriptors using DFT concepts characterize elements reactivity. Such descriptors, namely hardness and electrophilicity, are components of the derivative of the chemical potential. Their values form a new coordinates system, on which a third parameter can be mapped. The simplest mapping is the chemical potential itself, but other mapping may involve totally different chemical or physical parameters. Examples use rock analyses generated within the continental or oceanic crust of the Earth. They are usually described in an 11D system of major oxides. The new system of coordinates reduces the description to a more easily tractable 2D diagram. It also represents a base for plotting other chemical information, such as the normative component composition or a combination of them. Physically, other properties, such as the polymerization state or viscosity values, can be used to produce a 3D topography. Other topographic surfaces similar to the chemical potential of elements can be mapped, allowing quantification of partition coefficient values when elements fractionate in both liquid or viscous states. The reduction of an 11D diagram to a 2D one is suggested in other scientific descriptions of complex combinations.

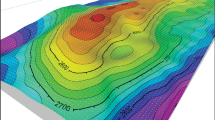
[ω-η] diagrams showing the chemical potential and the different continental and oceanic rock typesthen ading some chemical (Aluminium Saturation Index) parameter.















Similar content being viewed by others
References
Pearson RG (1963) Hard and soft acids and bases. J Am Chem Soc 85:3533–3539
Pearson RG (1988) Absolute electronegativity and hardness: application to inorganic chemistry. Inorg Chem 27:734–740
Chattaraj PK, Lee H, Parr RG (1991) HSAB principles. J Am Chem Soc 113:1855–1856
Pearson RG (2009) The hardness of closed systems. In: Chattaraj PK (ed) Chemical reactivity theory: a density functional view. CRC, Boca Raton, pp 155–163
Parr RG, Yang W (1984) Density functional approach to the frontier-electron theory of chemical reactivity. J Am Chem Soc 106:4049–4050
Chermette H (1999) Chemical reactivity indexes in density functional theory. J Comput Chem 20:129–154
Geerlings P, De Proft F, Langenaeker W (2003) Conceptual density functional theory. Chem Rev 103:1793–1873
Chattaraj PK, Maiti B (2004) Regioselectivity in the chemical reactions between molecules and protons: a quantum fluid density functional study. J Phys Chem A 10:658–664
Vigneresse JL, Duley S, Chattaraj PK (2011) Describing the chemical character of a magma. Chem Geol 287:102–113
Vigneresse JL (2012) Chemical reactivity parameters (HSAB) applied to magma evolution and ore formation. Lithos 153:154–164
McBirney AR (1993) Igneous petrology. Jones and Bartlett, Boston, p 572
Pearce JA, Harris NBW, Tindle AW (1984) Trace element discrimination diagrams for the tectonic interpretation of granitic rocks. J Petrol 25:956–983
Chayes F (1971) Ratio correlation a manual for students of petrology and geochemistry. Univ Chicago Press, Chicago, p 99
Pauling L (1932) The nature of the chemical bonds. IV The energy of single bonds and the relative electronegativity of atoms. J Am Chem Soc 54:3570–3582
Chattaraj PK, Giri S, Duley S (2012) Update 2 of: Electrophilicity index. Chem Rev 111:PR43–PR75
Ghanty TK, Gosh SK (1996) A density functional approach to hardness, polarizability, and valency of molecules in chemical reactions. J Phys Chem 100:12295–12298
Morell C, Grand A, Toro-Labbé A (2005) New dual descriptor for chemical reactivity. J PhysChem A109:205–212
Mortier WJ, Ghosh SK, Shankar S (1986) Electronegativity-equalization method for the calculation of atomic charges in molecules. J Am Chem Soc 108:4315–4320
Parr RG, Chattaraj PK (1991) Principle of maximum hardness. J Am Chem Soc 113:1854
Chattaraj PK (1996) The maximum hardness principle: an overview. Proc Indian Nat Sci Acad A62:513–531
Pan S, Sola M, Chattaraj PK (2013) On the validity of the maximum hardness principle and the minimum electrophilicity principle during chemical reactions. J Phys Chem A 117:1843–1852
Noorizadeh S, Shakerzadeh E (2008) A new scale of electronegativity based on electrophilicity index. J Phys Chem A 112:3486–3491
Morel C, Labet V, Grand A, Chermette H (2009) Minimum electrophilicity principle: an analysis based upon the variation of both chemical potential and absolute hardness. Phys Chem Chem Phys. 14:3417–3423
Torrent-Sucarrat M, Luis JM, Duran M, Sola M (2001) On the validity of the maximum hardness and minimum polarizability principles for nontotally symmetric vibrations. J Am Chem Soc 123:7951–7952
Wright S (1932) The roles of mutation, inbreeding, crossbreeding and selection in evolution proc. Sixth Ann Congr Genetics 1:356–366
Kauffman SA (1995) At home in the universe: the search for Laws of self-organization and complexity. Oxford University Press, New York 321 pp
Duley S, Vigneresse JL, Chattaraj PK (2012) Fitness landscapes in natural rocks system evolution: a conceptual DFT treatment. J Chem Sci 124:29–34
Das R, Vigneresse JL, Chattaraj PK (2014) Redox and Lewis acid–base activities through an electronegativity-hardness landscape diagram. J Mol Model 19:4857–4864
Mysen BO, Richet P (2005) Silicate glasses and melts. Elsevier, Amsterdam, p 544
Rollinson HR (1993) Using geochemical data: evaluation, presentation, interpretation. Longman Scientific and Technical, Harlow, p 352
Stacey FD, Banerjee SK (1974) The physical principles of rock magnetism. Elsevier, Amsterdam, p 204
Jugo PJ, Luth R, Richards J (2005) Experimental study of the sulfur content in basaltic melts saturated with immiscible sulfide or sulfate liquids at 1300 °C and 10 GPa. J Petrol 46:783–798
Jégo S, Dasgupta R (2014) The fate of sulfur during fluid-present melting of subducting basaltic crust at variable oxygen fugacity. J Petrol 55:1019–1050
Ishihara S (2004) The redox state of granitoids relative to tectonic setting and earth history: the magnetite–ilmenite series 30 years later. Trans R Soc Edinb Earth Sc 95:23–33
Ottonello G, Moretti R, Marini L, Vetuschi Zuccolini M (2001) Oxidation state of iron in silicate glasses and melts: a thermochemical model. Chem Geol 17:157–179
Candela PA (1989) Felsic magmas, volatiles, and metallogenesis. In: Whitney JA, Naldrett AJ (eds) Ore deposition associated with magmas. Rev Econ Geol 4:223–233
Blundy J, Wood B (2003) Partitioning of trace elements between crystals and melts. Earth Planet Sci Lett 210:383–397
Zhang Y, Ni H, Chen Y (2010) Diffusion data in silicate melts. Rev Min Geochem 72:311–408
Bodnar RJ (1995) Fluid-inclusion evidence for a magmatic source of metals in porphyry copper deposits. In: Thompson JFH (ed) Magmas, fluids, and ore deposits. Mineral Ass. Canada Short Course Series 23:139–52
Frezzotti ML (2001) Silicate-melt inclusions in magmatic rocks: applications to petrology. Lithos 55:273–299
Kamenetsky VS, Kamenestsky MB (2010) Magmatic fluids immiscible with silicate melts: examples from inclusions in phenocrysts and glasses, and implications for magma evolution and metal transport. Geofluids 10:293–311
Li Y, Audétat A (2012) Partitioning of V, Mn, Co, Ni, Cu, Zn, As, Mo, Ag, Sn, Sb, W, Au, Pb, and Bi between sulphide phases and hydrous basanite melt at upper mantle conditions. Geochim Cosmochim Acta 75:1673–1692
Zajacz Z, Candela PA, Piccoli PC, Sanchez-Valle C, Wälle M (2013) Solubility and partitioning behavior of Au, Cu, Ag and reduced S in magmas. Geochim Cosmochim Acta 112:288–304
Ghosh DC, Chakraborty T (2009) Gordy’s electrostatic scale of electronegativity revisited. J Mol Struct THEOCHEM 906:87–93
Atlan H (1989) Automata networks in immunology : their utility and their underdetermination. Bull Math Biol 54:247–253
Berg JM (2015) Biochemistry, W.H. Freeman, 1232 pp.
Atlan H (2018) Cours de Philosophie Biologiste et Cognitive. Odile Jacob (ed.), Paris, p 637
Acknowledgments
The paper came out after a stay at the Department of Chemistry, IIT Kharagpur, India, with granting by the CTS (Center for Theoretical Studies). It allowed fruitful introduction to DFT concepts and collaboration with Pratim K. Chattaraj and his students. Discussions with Christophe Morell (Université de Lyon1) encouraged me to formulate what is now this paper. Constructive reviews with comments are also warmly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Confict of interest
There is no conflict of interest.
Additional information
This paper belongs to Topical Collection International Conference on Systems and Processes in Physics, Chemistry and Biology (ICSPPCB-2018) in honor of Professor Pratim K. Chattaraj on his sixtieth birthday
Rights and permissions
About this article
Cite this article
Vigneresse, JL., Truche, L. Chemical descriptors for describing physico-chemical properties with applications to geosciences. J Mol Model 24, 231 (2018). https://doi.org/10.1007/s00894-018-3770-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-018-3770-0