Abstract
Background:
Multiple breath washout (MBW) is an informative but time-consuming test. This study evaluates the uncertainty of a time-saving predictor algorithm in adolescents.
Methods:
Adolescents were recruited from the Copenhagen Prospective Study on Asthma in Childhood (COPSAC2000) birth cohort. MBW trials were performed at 13 y of age with Innocor model Inn00400 using sulfur hexafluoride (SF6) as tracer gas. Measurements were analyzed using a mixed model focusing on two prediction points doubling (t5%) and quadrupling (t10%) the standard end point (t2.5%).
Results:
One hundred and seventy-two MBW trials conducted in 78 adolescents with and without asthma from COPSAC2000 were included. At t10%, the washout time (WoT) was reduced by 41%, and an uncertainty of 0.159 lung clearance index (LCI) units was introduced (±2 SD), ±1.27). At t5%, the WoT was reduced by 25%, with an uncertainty of 0.083 LCI units (±0.558). The optimal prediction point, which led to most saved time and least uncertainty was t5%.
Conclusion:
The predictor algorithm is capable of shortening the MBW test time but introduces an increasing uncertainty with earlier prediction points. This first-of-a-kind prediction algorithm holds promise in shortening the MBW test in children but should be used with caution in subjects with normal LCI values.
Similar content being viewed by others
Main
Multiple breath washout (MBW) is a promising tool for measuring lung function (1,2) requiring less cooperation, but longer measuring time than other methods, which highlights the need for time-saving procedures. The test investigates ventilation inhomogeneity of the lungs, which is currently measured as a lung clearance index (LCI). The MBW method used in this study measures LCI utilizing the nonresident tracer gas sulfur hexaflouride (SF6) requiring a wash-in phase prior to the washout phase. One such MBW trial lasts ~5 minutes, dependent on the age, body size, and lung status of the subject. According to Consensus guidelines, three trials are needed to complete a test making MBW testing rather time consuming (3).
MBW has been shown to be more sensitive than spirometry in detecting structural lung disease accompanying, e.g., cystic fibrosis (1). While spirometry requires active patient collaboration, MBW is measured during tidal breathing and may therefore be more feasible for children (4,5). However, some of the trials are typically interrupted due to impatience of the child. Additionally, changes in breathing pattern often occur at the end of the trial due to saliva production, dryness in the mouth, or lack of concentration, which can alter or even invalidate the test result. A reduction of test time therefore has the potential to increase success rate of the test by saving otherwise discarded trials, which could enable testing young children.
For historical reasons, the washout phase is proceeded until the end-tidal concentration of the tracer gas is 1/40 of the starting concentration (6). In this study, we used MBW data from adolescents participating in the Copenhagen Prospective Study on Asthma in Childhood (COPSAC2000) birth cohort to investigate the ability of a predictor algorithm to extrapolate LCI from higher end-tidal tracer gas concentrations. The aim was to evaluate the decrease in washout time (WoT) in relation to the introduced LCI uncertainty (LCI error (LCIe) at different points of prediction.
Results
Baseline
Seventy-eight (46% boys) of the 411 adolescents in the COPSAC2000 cohort were included in the study at mean (SD) age 13 y (0.64) and performed 216 trials. Fifty-four tests did not meet the predefined acceptability criteria and were excluded. Eighteen (21%) subjects had asthma at the time of measurement. Mean (SD) forced expiratory volume in 1 s (FEV1) / forced vital capacity (FVC) was 0.88 (0.07), mean (SD) functional residual capacity (FRC) was 1.93 l (0.45), and mean (SD) LCI was 6.22 units (0.51). Demographics, patient characteristics, and MBW baseline data are displayed in Table 1 . Number of individuals and included trials at each prediction point are displayed in Table 2 .
Error and Saved Time
In Figure 1 , the LCIe are shown with prediction limits and SDs. The lowest mean LCIe is found at t4% and the highest LCIe is found at t10%. At all prediction points, the mean error is positive, and the SD is increasing from t4% to t10%.
Mean prediction error (new – original) with 95% prediction limits in LCI units (left axis) at different prediction points. The column is decrease in washout time in seconds (right axis). LCI, lung clearance index; LPL,lower prediction limit; UPL,upper prediction limit.
In Table 3 , the decrease in WoT is shown at the different prediction points as saved time (ST). The lowest ST is found at t4% and the highest ST is found at t10%. In Table 1 , MBW data are shown with the coefficient of variation (CV) of FRC, original LCI (LCIOrg), LCI at t5% (LCINew5), and LCI at t10% (LCINew10). Median (interquartile range (IQR)) CV of LCIOrg is 3.2% (2.0;4.5), where median (IQR) CV for prediction point t5% and t10% is 4.7% (2.1;6.3) and 5.3% (2.3;9.5), respectively.
Prediction
The ST/highest prediction limit (HPL) ratio is highest at t5% indicating that this is the optimal extrapolation point ( Table 3 ).
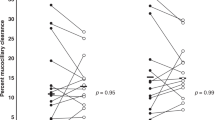
In Figure 2 , the difference between LCINew and LCIOrg is plotted against LCIOrg to examine for systematic errors in the algorithm. At t10%, the differences are distributed over a larger range than at t5%. At t5%, the distribution is not associated with the magnitude of the mean LCIOrg, whereas at t10%, the distribution seems more scattered in the higher values of LCI (7 to 8), with fewer observations near zero. However, logarithmic transformation of the differences did not improve the distribution (data not shown).
Bland–Altman plot for prediction points (a) t5% and (b) t10%, doubling and quadrupling the standard prediction point of t2.5%, i.e., 1/40 of the starting concentration. The difference between predicted (new) and original (org) data is plotted against original result.
When extrapolating from t10%, we found a mean LCIe (±2 SD) of 0.159 (±1.27) and a mean ST of 38.6 s. In comparison, when extrapolating from t5%, we found a mean LCIe of 0.083 (±0.558) and a mean ST of 23.6 s. At t5% where ST/HPL is highest, the LCIe (±2SD) equals 1.3% (±9%) of the mean LCI and the WoT is reduced with 25.3%.
Discussion
The proposed predictor algorithm is capable of shortening the MBW test procedure and provides an estimate of the LCI with a mean LCIe (±2SD) of 1.3% (±9%) and a reduction in mean WoT of 25.3% at the optimal prediction point.
At all prediction points the mean LCIe is positive, and the SD is increasing from t4% to t10%. This implies that the algorithm is overestimating LCI and indicates that earlier prediction points increase the uncertainty. To determine the optimal prediction point to use for extrapolation, it is relevant to evaluate the amount of saved time in relation to the LCIe and the SD of this. HPL gives an idea of the most extreme error the algorithm introduces, and from our data, a direct correlation of the saved time and HPL suggests that the optimal point of prediction is t5%. At this prediction point, the HPL is 0.64 LCI units, which is considered a high inaccuracy in subjects with normal LCI values.
The median CV for LCIOrg in our study was 3.2%, which is comparable to other studies reporting CV for LCIOrg from 4 to 9% (7,8,9,10). This implies that the MBW measurement itself is affected by some uncertainty, which should be taken into account when evaluating the algorithm. The CV is higher for extrapolated LCI values compared to the CV of LCIOrg, underlining the uncertainty introduced by the algorithm.
Lung function measurements are commonly used in a clinical setting to distinguish between normal and abnormal subjects and to monitor disease progressing. Our study was cross-sectional, why we cannot evaluate how the algorithm performs over time in the same subject.
Shortening of the MBW test time has been the topic of interest of other studies. Yammine et al. (11) examined sensitivity and specificity in children with cystic fibrosis when stopping the washout at 5% of the starting concentration. They found a similar or improved sensitivity and specificity with this prediction point compared with the traditional end point of 2.5% and proposed this as a way of shortening test time. The average of saved measuring time in cystic fibrosis patients was 30 vs. 21% in controls (11), which is comparable to our findings of a 25% reduction of WoT in a mixed cohort of asthmatics and healthy adolescents.
The predictor algorithm is capable of shortening the washout phase in each trial by 20–30 s when extrapolating from t5%. This may be considered negligible but actually accounts for at least a 25% reduction of the washout phase and 10–15% reduction of the entire test time, which may facilitate and ease testing of young children. Moreover, the wash-in phase will also be shortened, as the concentration of tracer gas at the beginning of the wash-in will be higher.
The algorithm, which is based on washout of SF6 tracer gas, approved for use with Innocor (Innovision ApS, Glamsbjerg, Denmark) in the European Union and the United States, could be modified to include washout of other tracer gasses such as N2.
In conclusion, this study investigates a new approach to the MBW method using a novel predictor algorithm with the potential to save test time and avoid discarded tests in impatient children. The algorithm is capable of shortening the test with at least 10–15% with a mean LCIe (±2 SD) of 1.3% (±9%) but introduces a potential uncertainty of around 0.6 LCI units.
Methods
Study Population
Participants in this study were recruited from the COPSAC2000 cohort (12), which is a prospective, longitudinal, birth-cohort study of 411 infants born to asthmatic mothers in 1998–2001. The children were enrolled at 1 mo of age excluding children with severe congenital abnormality, gestational age <36 wk, and any lung symptoms prior to enrolment. The children were followed closely from birth with scheduled clinic visits every half-year until age 7 y and again at age 13 y. All the MBW measurements utilized in this study were conducted routinely during the scheduled 13-y visit at the COPSAC clinic. The 78 included participants were the ones who had completed MBW measurements by the time of data analysis.
Lung symptoms were recorded in daily diaries fulfilled from birth by the parents. Asthma was defined according to a strict predefined algorithm based on these lung symptom recordings and response to treatment and was exclusively diagnosed by the pediatricians working at the COPSAC clinic. Adolescents without asthma were defined as healthy in this study.
MBW Measurements
The Innocor model Inn00400, (Innovision ApS), which was used to perform MBW assessments in this study, was validated as described by Gonem et al. (13). It was calibrated on a daily basis according to manufacturer’s guidelines and used an almost fully automatic software that ensured an equal measuring technique independent of the observer. The device has a re-breathing valve unit with connections to a re-breathing bag for the wash-in phase of the tracer gas SF6, a flow meter connected to a mouthpiece via a bacterial filter, and room air. MiniValve with a pre-capillary dead space of 62.3 ml was inserted in the re-breathing valve unit. The mean (SD) weight of the participants was 52.0 kg (10.6) giving a mean pre-capillary dead space per kg body weight (SD) of 1.25 ml/kg (0.24).
For the wash-in phase, the device supplied a gas mixture containing 0.2% SF6 and 35.5% oxygen (O2) among others and used a scrubber to clear the produced carbon dioxide (CO2). Participants wore noseclips and breathed through mouthpieces (small, 22 mm, Hans Rudolph, Shawnee, KS) attached to a flow meter (4719 series Hans Rudolph) until gas equilibrium was reached, i.e., the variation of the SF6 concentration between inspiration and expiration was below 2%. The connections switched from re-breathing bag to room air at the first exhalation of the washout phase. The participants breathed room air until the end tidal concentration of SF6 had reached a level below 1/40th of the starting concentration in three consecutive breaths. Only successfully finished trials were included. The outcomes that were used in the analysis were LCI, FRC, and WoT.
The collected data were sent to Innovision ApS, where the algorithm was used to extrapolate LCI values. Thereafter, raw data and the extrapolated values were sent to the COPSAC clinic, where the statistical analysis was performed, and the results were interpreted. Analysis was focused on two prediction points doubling (t5%) and quadrupling (t10%) the standard finish point of the trial (2.5% of the initial tracer gas concentration).
Predictor Algorithm
The algorithm was designed to predict the LCI and FRC at 2.5% (1/40) of the starting concentration at prediction points between 10 and 4% of the starting concentration. The washout part of SF6 was first normalized to an even breathing tidal volume using the phase three slope of the SF6. Then, a best-fit-curve through the last nine end-tidal values was extrapolated to the normal 2.5%, and the estimated expired cumulated volume (VCE) was found. A similar best-fit-curve was done on the SF6 volume of each breath and by extrapolating to the estimated VCE the total volume of expired SF6 was estimated, and the FRC was determined. Finally, the predicted LCI was calculated as the estimated VCE divided by the estimated FRC.
Statistics
A predefined acceptability criterion was made for MBW trials in the same subject: (FRCx − FRCmean)/FRCmean <4.8%, which is in agreement with the most narrow acceptability criterion of ERS/ATS consensus statement (3). If all trials from an individual varied >4.8%, the individual was excluded.
LCIe is defined as the difference between LCINew and LCIOrg (LCINew − LCIOrg). ST is defined by the difference between the original WoT and the time at prediction. The HPL is the numerical value of LCIe plus 2 SD. The ratio ST/HPL is calculated by dividing saved time with HPL. It is used to find the optimal prediction point regarding the amount of saved time in relation to the size of the error obtained when using the algorithm.
Individual CV was calculated by the SD of all trials of the individual divided by the mean. A median CV and IQR were calculated for LCIOrg, FRC, LCINew5, and LCINew10, as the values were not normally distributed.
Measurements of LCIe and ST were analyzed using a mixed model with the individual as a random effect in order to take account of the repeated measurements on the same individual. Mean values for LCIe and ST were calculated as the overall estimate of the intercept in this model. These were compared to the mean LCI and mean WoT to calculate the percentage deviation and reduction. For the LCIe, SDs of the individual observations were calculated by summing up between- and within-individual variance using mixed model estimates. Prediction limits were defined as the 95% confidence limits on the individual observations calculated using the SD for individual observations. For saved time, SE of the mean saved time is provided by estimating the SE of the intercept in the mixed model.
Prediction at t5% and t10% were chosen for further analyses as the concentrations are two and four times the concentration at the end of a conventional test (1/40 of the starting concentration), respectively.
Prediction point-dependent LCIe was assessed using plots showing mean and upper and lower prediction limits. Prediction point-dependent saved time was compared to LCIe. Agreement between LCIOrg and LCINew, thus the LCIe, was assessed using Bland–Altman plots in t5% and t10% with limits of agreement (mean ± 2SD) shown as dotted lines (14).
All statistics on algorithm data were carried out using SAS version 9.3 and SAS Enterprise Guide 5.1 version 5.100.0.12019 (SAS, Cary, NC) at the COPSAC clinic.
Ethics
The Copenhagen Ethics Committee (KF 01-289/96) and The Danish Data Protection Agency (2008-41-2434) approved the study, and informed consent was obtained from both parents at enrollment.
Statement of Financial Support
COPSAC is funded by private and public research funds all listed on www.copsac.com. The Lundbeck Foundation, Copenhagen, Denmark; Danish State Budget, Copenhagen, Denmark; Danish Council for Strategic Research, Copenhagen, Denmark; The Danish Council for Independent Research, Copenhagen, Denmark; and The Capital Region Research Foundation, Hillerød, Denmark, have provided core support for COPSAC. The funding agencies did not have any influence on study design, data collection, data analysis, decision to publish, or preparation of the manuscript.
Disclosure
The authors declare no conflict of interests.
References
Gustafsson PM, De Jong PA, Tiddens HA, Lindblad A. Multiple-breath inert gas washout and spirometry versus structural lung disease in cystic fibrosis. Thorax 2008;63:129–34.
Macleod KA, Horsley AR, Bell NJ, Greening AP, Innes JA, Cunningham S. Ventilation heterogeneity in children with well controlled asthma with normal spirometry indicates residual airways disease. Thorax 2009;64:33–7.
Robinson PD, Latzin P, Verbanck S, et al. Consensus statement for inert gas washout measurement using multiple- and single- breath tests. Eur Respir J 2013;41:507–22.
Gustafsson PM. Inert gas washout in preschool children. Paediatr Respir Rev 2005;6:239–45.
Beydon N, Davis SD, Lombardi E, et al.; American Thoracic Society/European Respiratory Society Working Group on Infant and Young Children Pulmonary Function Testing. An official American Thoracic Society/European Respiratory Society statement: pulmonary function testing in preschool children. Am J Respir Crit Care Med 2007;175:1304–45.
Robinson PD, Goldman MD, Gustafsson PM. Inert gas washout: theoretical background and clinical utility in respiratory disease. Respiration 2009;78:339–55.
Zwitserloot A, Fuchs SI, Müller C, Bisdorf K, Gappa M. Clinical application of inert gas multiple breath washout in children and adolescents with asthma. Respir Med 2014;108:1254–9.
Amin R, Subbarao P, Jabar A, et al. Hypertonic saline improves the LCI in paediatric patients with CF with normal lung function. Thorax 2010;65:379–83.
Fuchs SI, Eder J, Ellemunter H, Gappa M. Lung clearance index: normal values, repeatability, and reproducibility in healthy children and adolescents. Pediatr Pulmonol 2009;44:1180–5.
Aurora P, Gustafsson P, Bush A, et al. Multiple breath inert gas washout as a measure of ventilation distribution in children with cystic fibrosis. Thorax 2004;59:1068–73.
Yammine S, Singer F, Abbas C, Roos M, Latzin P. Multiple-breath washout measurements can be significantly shortened in children. Thorax 2013;68:586–7.
Bisgaard H. The Copenhagen Prospective Study on Asthma in Childhood (COPSAC): design, rationale, and baseline data from a longitudinal birth cohort study. Ann Allergy Asthma Immunol 2004;93:381–9.
Gonem S, Singer F, Corkill S, Singapuri A, Siddiqui S, Gustafsson P. Validation of a photoacoustic gas analyser for the measurement of functional residual capacity using multiple-breath inert gas washout. Respiration 2014;87:462–8.
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986;1:307–10.
Author information
Authors and Affiliations
Corresponding author
PowerPoint slides
Rights and permissions
About this article
Cite this article
Grønbæk, J., Hallas, H., Arianto, L. et al. New time-saving predictor algorithm for multiple breath washout in adolescents. Pediatr Res 80, 49–53 (2016). https://doi.org/10.1038/pr.2016.57
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pr.2016.57
This article is cited by
-
Feasibility and clinical applications of multiple breath wash-out (MBW) testing using sulphur hexafluoride in adults with bronchial asthma
Scientific Reports (2020)
-
Multiple breath washout testing in adults with pulmonary disease and healthy controls – can fewer measurements eventually be more?
BMC Pulmonary Medicine (2017)