Abstract
Purpose
This study assesses the potential of two contrasted fragrant Pelargonium cultivars to induce pH and dissolved organic carbon (DOC) changes in the soil solution, Pb speciation, and their subsequent effects on rhizosphere phytoavailable Pb.
Materials and methods
Rooted plantlets were grown in special devices, floating on aerated nutrient solution in PVC tanks. This setup allows roots to be physically separated, through a mesh, from a 3-mm soil matrix layer that can be considered as rhizosphere soil. Two contrasted soils, each spiked with Pb-rich particles, emitted from a battery recycling industry, were used at total burdens of 500 and 1500 mg Pb kg−1 in addition to a control unspiked soil. Soil solution pH, phytoavailable Pb, DOC, Pb adsorption, precipitation on roots, and Pb phases in soil and plant were investigated.
Results and discussion
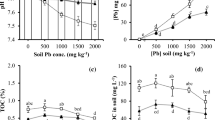
Attar of Roses (Attar) cultivar acidified its rhizosphere by 0.4 pH units in both spiked soils. Concolor Lace (Concolor) was unable to change soil solution pH on soil-1 and increased it by 0.7 units on soil 2. Concentrations of Pb in soil solution from Attar plants were always higher than those of Concolor ones. DOC contents of both unspiked soil-1 and soil-2 without plants were not significantly different. In the case of spiked samples, DOC contents in the rhizosphere soil were increased by three and two times for Attar and Concolor, respectively, compared to the unspiked soil without plant. Both cultivars were able to increase DOC contents, independent of soil type and level of contamination. Accumulation of Pb in shoots and roots was higher in Attar as compared to Concolor due to enhanced available Pb as a result of pH and DOC modifications of the rhizosphere soil. Significant amounts of Pb were adsorbed on roots of both cultivars. X-ray elemental analysis of precipitates on roots revealed the association of Pb with P in cylinder-like structures. Extended X-ray absorption fine structure (EXAFS) spectroscopy revealed that Pb was present, to a major extent in the inorganic form, mainly as PbSO4 in the soil, whereas it was complexed with organic species within plant tissues. The conversion of Pb into organic species could decrease toxicity, may enhance plant tolerance, and could increase translocation.
Conclusions
Plant-induced changes were responsible for the modification of lead phases within the soil. Immobile forms present in the source leaded particles as well as in the soils were converted into soluble species, ultimately improving the phytoavailable or soil solubilized Pb.





Similar content being viewed by others
References
Angeles OR, Johnson SE, Buresh RJ (2006) Soil solution sampling for organic acids in rice paddy soils. Soil Sci Soc Am J 70:48–56
Arshad M, Silvestre J, Pinelli E, Kallerhoff J, Kaemmerer M, Tarigo A, Shahid A, Guiresse M, Pradere P, Dumat C (2008) A field study of lead phytoextraction by various scented pelargonium cultivars. Chemosphere 71:2187–2192
Belin S, Briois V, Traverse A, Idir M, Moreno T, Ribbens M (2005) SAMBA a new beamline at SOLEIL for x-ray absorption spectroscopy in the 4–40 keV energy range. Phys Scr T115:980–983
Cabala J, Teper L (2007) Metalliferous constituents of rhizosphere soils contaminated by Zn-Pb mining in southern Poland. Water Air Soil Pollut 178:351–362
Cecchi M, Dumat C, Alric A, Felix-Faure B, Pradere P, Guiresse M (2008) Multi-metal contamination of a calcic cambisol by fallout from a lead-recycling plant. Geoderma 144:287–298
Chaignon V, Hinsinger P (2003) A biotest for evaluating copper bioavailability to plants in a contaminated soil. J Environ Qual 32:824–833
Chen B, Shan XQ, Qian J (1996) Bioavailability index for quantitative evaluation of plant availability of extractable soil trace elements. Plant Soil 186:275–283
Cheng S-F, Huang C-Y, Lin Y-C, Lin S-C, Chen K-L (2015) Phytoremediation of lead using corn in contaminated agricultural land–An in situ study and benefit assessment. Ecotox Environ Saf 111:72–77
Cotter-Howells JD, Champness PE, Charnock JM (1999) Mineralogy of Pb-P grains in the roots of Agrostis capillaris L. by ATEM and EXAFS. Mineral Mag 63:777–789
Dixit R, Wasiullah MD, Kuppusamy P, Singh UB, Sahu A, Shukla R, Singh BP, Rai JP, Sharma PK, Lade H, Paul D (2015) Bioremediation of heavy metals from soil and aquatic environment: an overview of principles and criteria of fundamental processes. Sustainability 7:2189–2212
FAO (1998) World reference base for soil resources. Food and Agriculture Organization of the United Nations, Rome
Fernández R, Bertrand A, García JI, Tamés RS, González A (2012) Lead accumulation and synthesis of non-protein thiolic peptides in selected clones of Melilotus alba and Melilotus officinalis. Environ Exp Bot 78:18–24
Ferrand E, Dumat C, Leclerc-Cessac E, Benedetti MF (2006) Phytoavailability of zirconium in relation to its initial added form and soil characteristics. Plant Soil 287:313–325
Hansel CM, Fendorf S, Sutton S, Newville M (2001) Characterization of Fe plaque and associated metals on the roots of mine-waste impacted aquatic plants. Environ Sci Technol 35:3863–3868
Kabata-Pendias A (2004) Soil-plant transfer of trace elements-an environmental issue. Geoderma 122:143–149
Khan S, Cao Q, Chen BD, Zhu YG (2006) Humic acids increase the phytoavailability of Cd and Pb to wheat plants cultivated in freshly spiked, contaminated soil. J Soils Sediments 6:236–242
Kidd PS, Monterroso C (2005) Metal extraction by Alyssum serpyllifolium ssp lusitanicum on mine-spoil soils from Spain. Sci Total Environ 336:1–11
Maestri E, Marmiroli M, Visioli G, Marmiroli N (2010) Metal tolerance and hyperaccumulation: costs and trade-offs between traits and environment. Environ Exp Bot 68:1–13
Manceau A, Boisset MC, Sarret G, Hazemann JL, Mench M, Cambier P, Prost R (1996) Direct determination of lead speciation in contaminated soils by EXAFS spectroscopy. Environ Sci Technol 30:1540–1552
Meyers DER, Kopittke PM, Auchterlonie GJ, Webb RI (2009) Characterization of lead precipitate following uptake by roots of Brassica juncea. Environ Toxicol Chem 28:2250–2254
Pendergrass A, Butcher DJ (2006) Uptake of lead and arsenic in food plants grown in contaminated soil from Barber Orchard, NC. Microchem J 83:14–16
Rascio N, Navari-Izzo F (2011) Heavy metal hyperaccumulating plants: how and why do they do it? And what makes them so interesting? Plant Sci 180:169–181
Ravel B, Newville M (2005) ATHENA and ARTEMIS: interactive graphical data analysis using IFEFFIT. J Synchrotron Radiat 12:537–541
Sarret G, Manceau A, Cuny D, Haluwyn CV, Deruelle S, Hazemann JL, Soldo Y, Eybert-Bérard L, Menthonnex JJ (1998a) Mechanism of lichen resistance to metallic pollution. Environ Sci Technol 32:3325–3330
Sarret G, Manceau A, Spadini L, Roux JC, Hazemann JL, Soldo Y, Eybert-Bérard L, Menthonnex JJ (1998b) EXAFS determination of Pb, Zn complexing sites of Penicillium chrysogenum cell walls. Environ Sci Technol 32:1648–1655
Sauve S, Dumestre A, McBride M, Hendershot W (1998) Derivation of soil quality criteria using predicted chemical speciation of Pb2+ and Cu2+. Environ Toxicol Chem 17:1481–1489
Shahid M, Arshad M, Kaemmerer M, Pinelli E, Probst A, Baque D, Pradere P, Dumat C (2012) Long-term field metal extraction by Pelargonium: phytoextraction efficiency in relation to plant maturity. Int J Phytorem 14:493–505
Sorvari J (2007) Environmental risks at Finnish shooting ranges – a case study. Hum Ecol Risk Assess 13:1111–1146
Straczek A, Sarret G, Manceau A, Hinsinger P, Geoffroy N, Jaillard B (2008) Zinc distribution and speciation in roots of various genotypes of tobacco exposed to Zn. Environ Exp Bot 63:80–90
Strømseng AE, Ljønes M, Bakka L, Mariussen E (2009) Episodic discharge of lead, copper and antimony from a Norwegian small arm shooting range. J Environ Monitor 11:1259–1267
Sungur A, Soylak M, Ozcan H (2014) Investigation of heavy metal mobility and availability by the BCR sequential extraction procedure: relationship between soil properties and heavy metals availability. Chem Speciat Bioavailab 26:219–230
Uzu G, Sobanska S, Aliouane Y, Pradere P, Dumat C (2009) Study of lead phytoavailability for atmospheric industrial micronic and sub-micronic particles in relation with lead speciation. Environ Pollut 157:1178–1185
Uzu G, Sobanska S, Sarret G, Munoz M, Dumat C (2010) Foliar lead uptake by lettuce exposed to atmospheric fallouts. Environ Sci Technol 44:1036–1042
Uzu G, Sobanska S, Sarret G, Sauvain JJ, Pradere P, Dumat C (2011) Characterization of lead-recycling facility emissions at various workplaces: major insights for sanitary risks assessment. J Hazard Mater 186:1018–1027
Wang G, Su MY, Chen YH, Lin FF, Luo D, Gao SF (2006) Transfer characteristics of cadmium and lead from soil to the edible parts of six vegetable species in southeastern China. Environ Pollut 144:127–135
Wang HH, Shan XQ, Liu T, Xie YN, Wen B, Zhang SZ, Han F, van Genuchten MT (2007) Organic acids enhance the uptake of lead by wheat roots. Planta 225:1483–1494
Acknowledgments
The authors would like to thank the Higher Education Commission of Pakistan (www.hec.gov.pk) for sponsoring Muhammad Arshad. The Society of Chemical Treatment of Metals (STCM, http://www.stc-metaux.com/) is acknowledged for providing Pb particles used in the experiments. We also thank Dr. Sohail Ejaz, University of Cambridge, UK, for constructive proof reading and English language corrections.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Dong-Mei Zhou
Rights and permissions
About this article
Cite this article
Arshad, M., Merlina, G., Uzu, G. et al. Phytoavailability of lead altered by two Pelargonium cultivars grown on contrasting lead-spiked soils. J Soils Sediments 16, 581–591 (2016). https://doi.org/10.1007/s11368-015-1248-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-015-1248-6